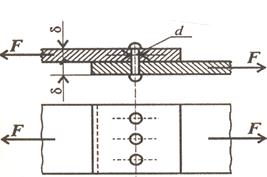
Conductivity and superconductivityThe electrical conductivity of a metal (or its reciprocal, electrical resistivity) is determined by the ease of movement of electrons past the atoms under the influence of an electric field. This movement is particularly easy in copper, silver, gold, and aluminium. The conductivity of a given metal is decreased by phenomena that deflect, or scatter, the moving electrons. These can be anything that destroys the local perfection of the atomic arrangement—for example, impurity atoms, grain boundaries, or the random oscillation of atoms induced by thermal energy. This is why the conductivity of a metal increases substantially with falling temperature: in a pure metal at room temperature, most resistance to the motion of free electrons comes from the thermal vibration of the atoms; if the temperature is reduced to almost absolute zero, where thermal motion essentially stops, conductivity can increase several thousandfold. Superconductivity was first discovered by Dutch physicist Heike Kamerlingh Onnes (1853–1926) in 1911. While studying the properties of materials near absolute zero (0 K, − 273°C, or − 459°F), Kamerlingh Onnes found that some materials lose all resistance to the flow of electric current at these temperatures. The applications for superconducting materials fall into two general categories: electronics and magnets. All electronic devices will operate more efficiently if they are made from superconducting materials. However, those materials have to be kept at low temperature. Those applications have only a limited commercial application so far. The situation is very different with magnets. The most powerful magnets are electromagnets —magnets that owe their magnetic properties to the flow of electric current through a metal core. The traditional way to make a more powerful magnet is to make the metal core larger and larger. The problem with this approach, is that the core needed to make very powerful magnets is larger than can be used on a practical basis. Using superconducting materials, however, the flow of electric current is more efficient, and a more powerful magnet can be made with a smaller metal core. 6. Проверьте усвоенную информацию ответив на следующие вопросы на английском языке: 1. What metals easily conduct electricity? 2. How do impurity atoms and grain boundaries affect the flow of electrons? 3. Why does conductivity of most metals increases with falling temperature? 4. Where are superconducting materials applied? 7. Дайте как можно больше производных от следующих слов, добавляя подходящие суффиксы: Например: add addition additional additionally additive differ combine act identify practice special equip procedure investigate pure afford expand solve attract complete extend pollute distribute conduct classify resist vary apply consider strong probable resistance use apply
|